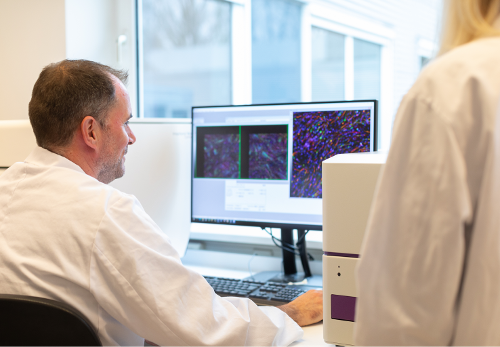
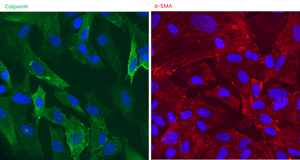
Perfecting the full potential of iPSCs to transform drug discovery in human healthcare
We enable you to make the most confident decisions for your preclinical candidates as early as possible
Increase Confidence in the Discovery of Breakthrough Therapeutics
For more than a decade, Ncardia has partnered with biotech and pharmaceutical organizations to tackle one of the biggest challenges in drug discovery: even after enormous investments of time and resources, 9 out of 10 drugs fail the first time they contact a human.
You can dramatically improve those odds – using an integrated iPSC-based drug discovery platform that allows you to mimic drug interactions with the human body from hit identification to lead optimization.
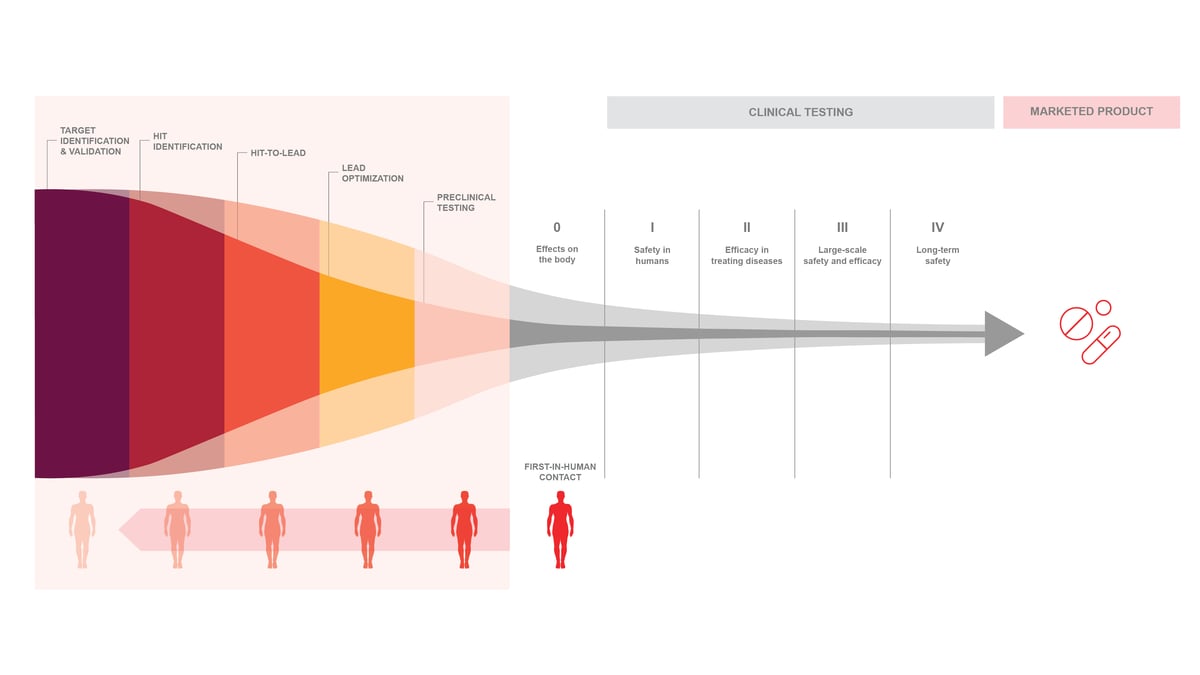
Proven, integrated iPSC-platform to
support multi-year drug discovery programs
By continually cultivating a special expertise in human biology,
Ncardia has established a portfolio of relevant cell models and screening assays
that primes your team for a higher probability of downstream
success to help avoid costly attrition.
Advancing drug discovery with human iPSC technology
Ncardia has the experience, technical expertise and strategic insight to help you fail faster – eliminating non-valuable compounds at the earliest stages of drug discovery and enabling you to quickly and confidently focus on the most promising candidates.
companies served with
iPSC projects in 2020-21
projects we’ve supported
since 2020
from distinct sources
pursuing iPSC-based
innovations
Use the power of our iPSC platform to catalyze your discovery programs
As more and more drug discovery innovators look to capitalize on the unique value only iPSCs can deliver, they look to Ncardia as the uniquely qualified collaborator to join them on their all-important journeys. We’re ready to help make your next step the very best it can be. So let’s start with a conversation – about your vision, goals and expectations for your drug discovery projects.